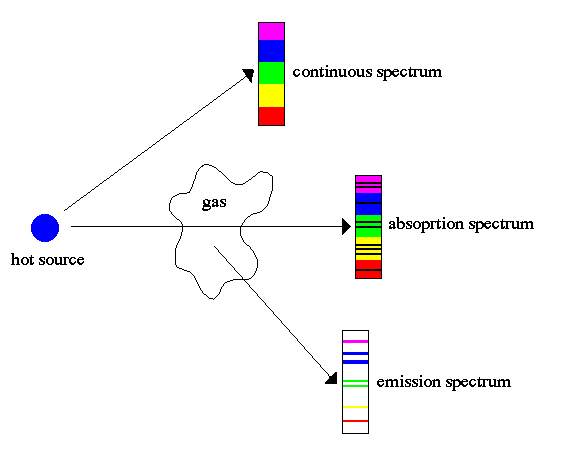
Absorbance Spectroscopy, Absorbance Explained
In absorbance spectroscopy (also known as absorption spectroscopy), a spectrometer is used to directly measure how much light is absorbed by a sample as a function of wavelength. Quantifying the absorbance of light by an atom or molecule can provide important information about the molecules electronic structure. Depending on the sample, absorbance measurements can also give you key insights into other material properties, such as sample concentration, phase changes, or composition changes. Spectrometer and Accessories Low Price Optical Spectrometer Light Sources And More Complete Kits for £2,400 Explore Now Contents Absorbance Theory Measuring Absorbance Defining Absorbance Molar Attenuation Coefficient Absorption Coefficient Absorbance Units Absorbance Uses Resources and References Absorbance Theory Band theory describes how electrons are organised within a solid. This can be useful when discussing different properties of a solid (conductors, semiconductors, or metals). Within an atom, electrons can exist in regions around
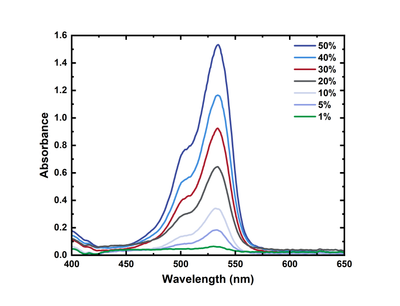
ACTTR Inc. - Understanding Spectrophotometer (4) Quantitative Analysis
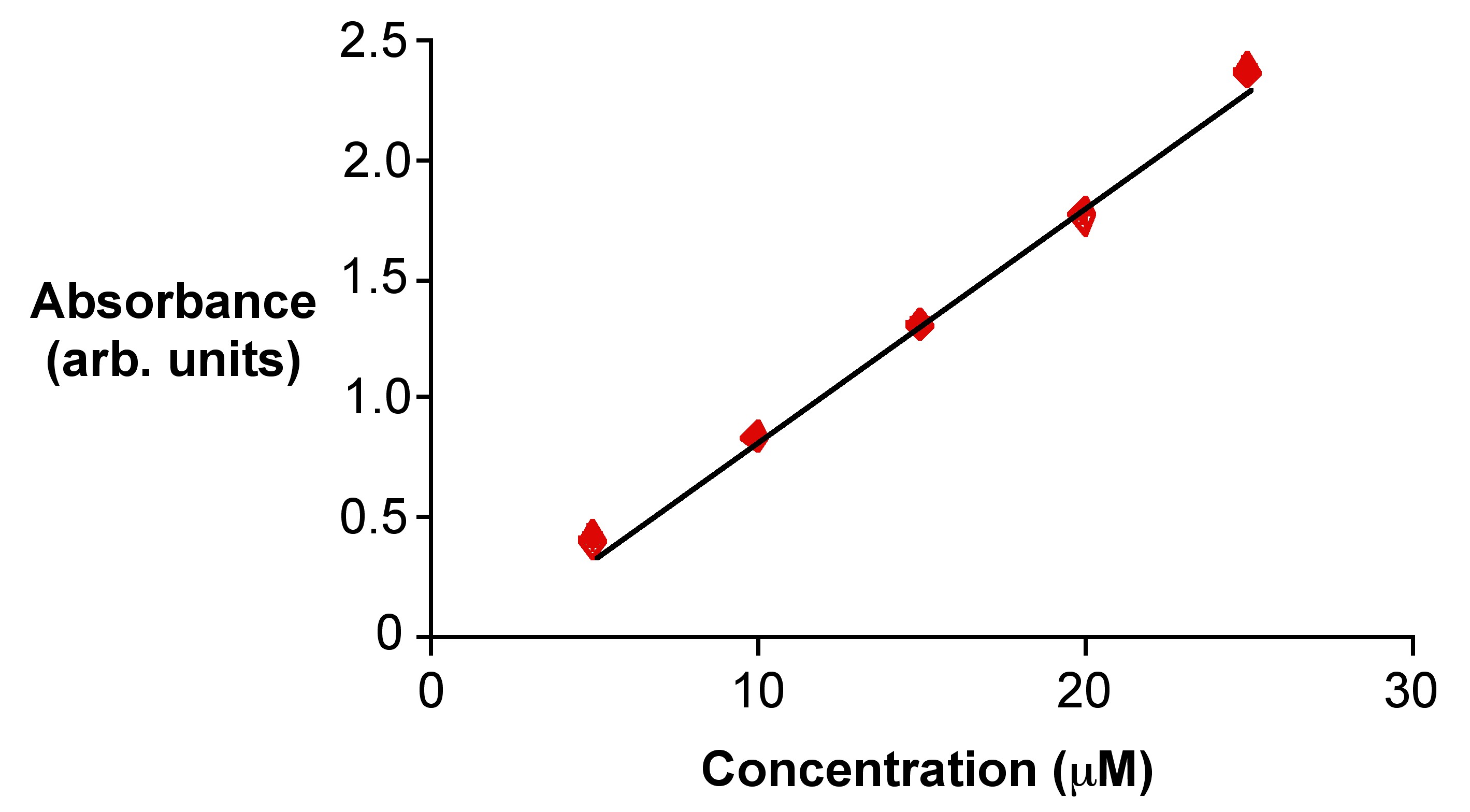
4.4: UV-Visible Spectroscopy - Chemistry LibreTexts
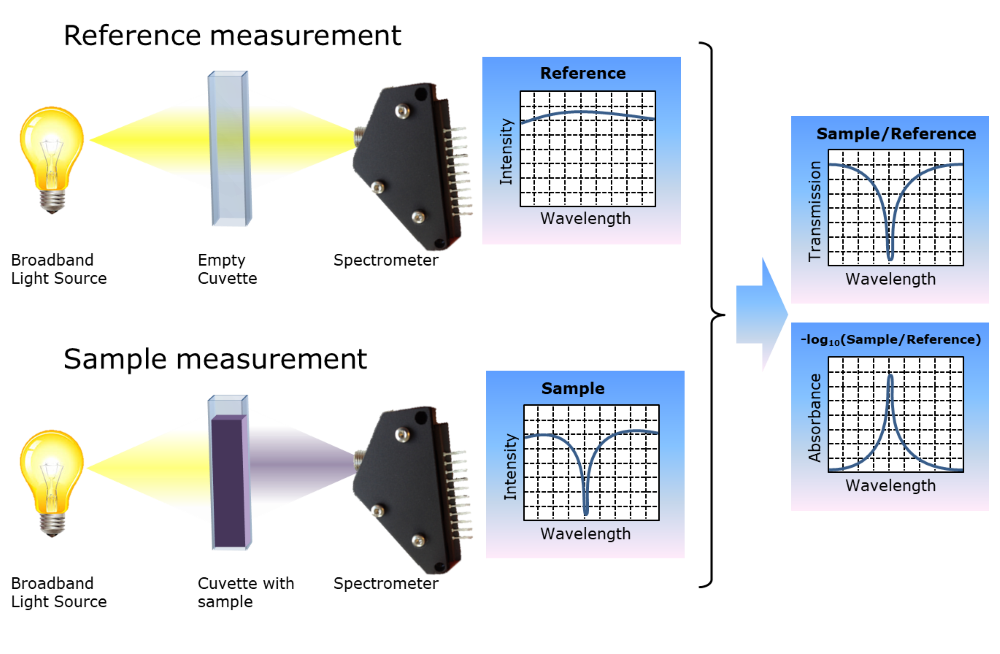
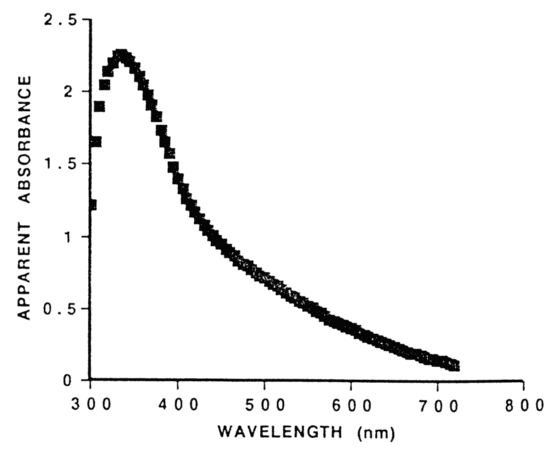
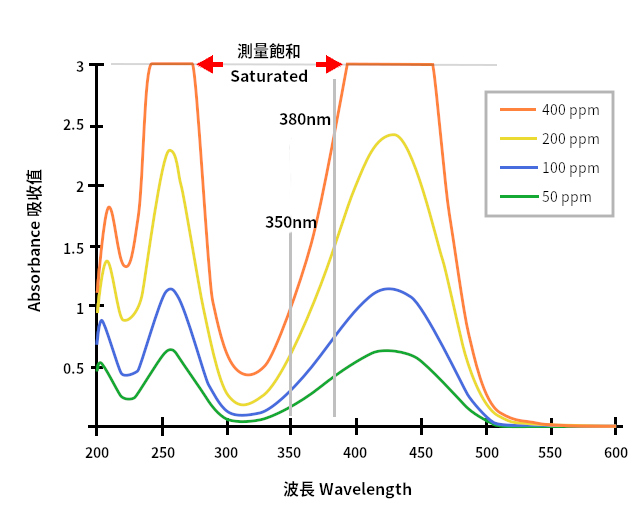
How to Obtain a Large Absorbance Range - Ibsen Photonics

Atomic Absorption Spectroscopy, Principles and Applications

atomic absorption spectroscopy
Absorption band - Wikipedia

Absorbance Spectroscopy, Absorbance Explained

Molecular Absorption - an overview

How to calculate the energy that an electron absorbed from one
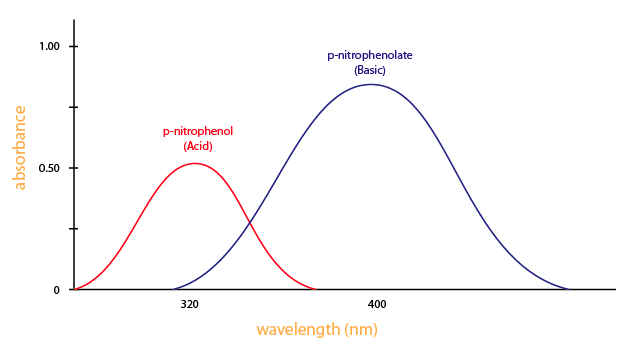
2.1.5: Spectrophotometry - Chemistry LibreTexts
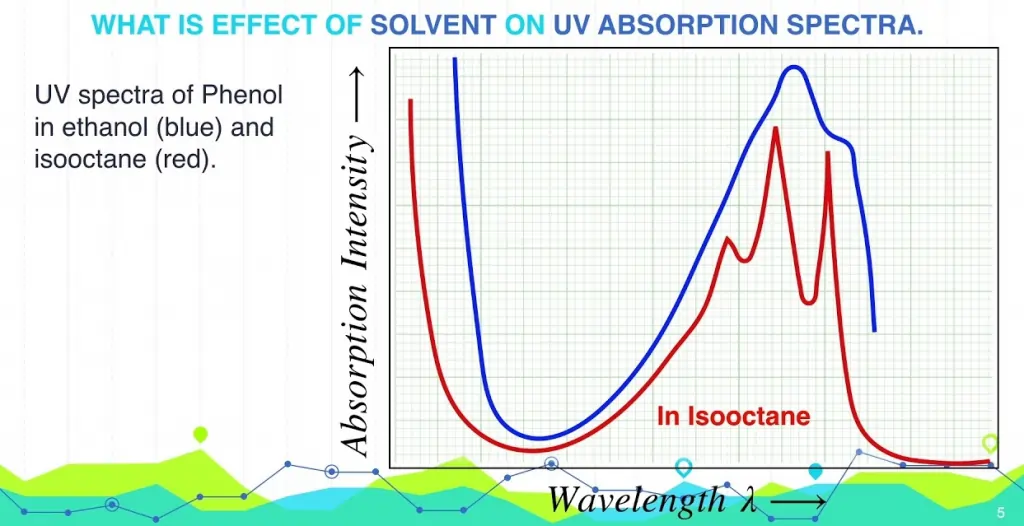
Nature of Factors Impacting UV-Vis Spectroscopy

UV-Vis Spectroscopy: Principle, Strengths and Limitations and
What is UV-Vis Spectroscopy? And How Does It Apply To Conjugation?
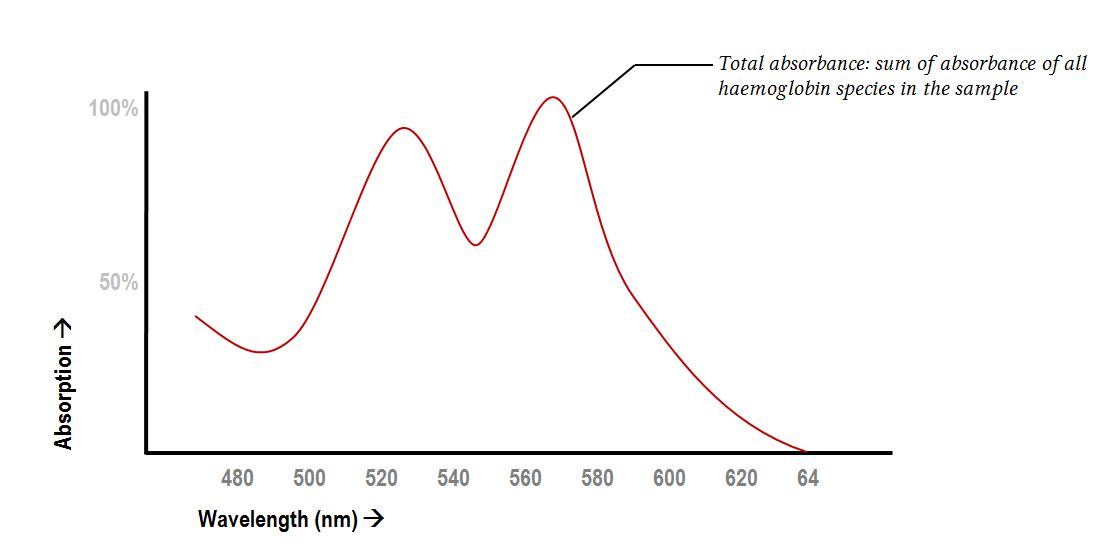
Absorption spectroscopy of haemoglobin species

Absorption spectroscopy of haemoglobin species