Emission & Absorption Spectrum: Emission,Absorption,Spectroscopy ,Line Spectrum
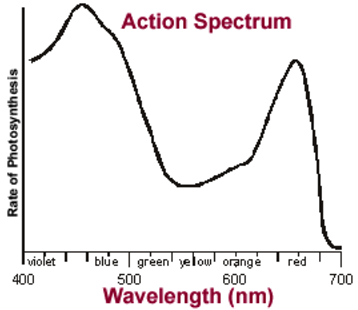
The spectrum of radiation emitted by a substance that has absorbed energy is called an emission spectrum

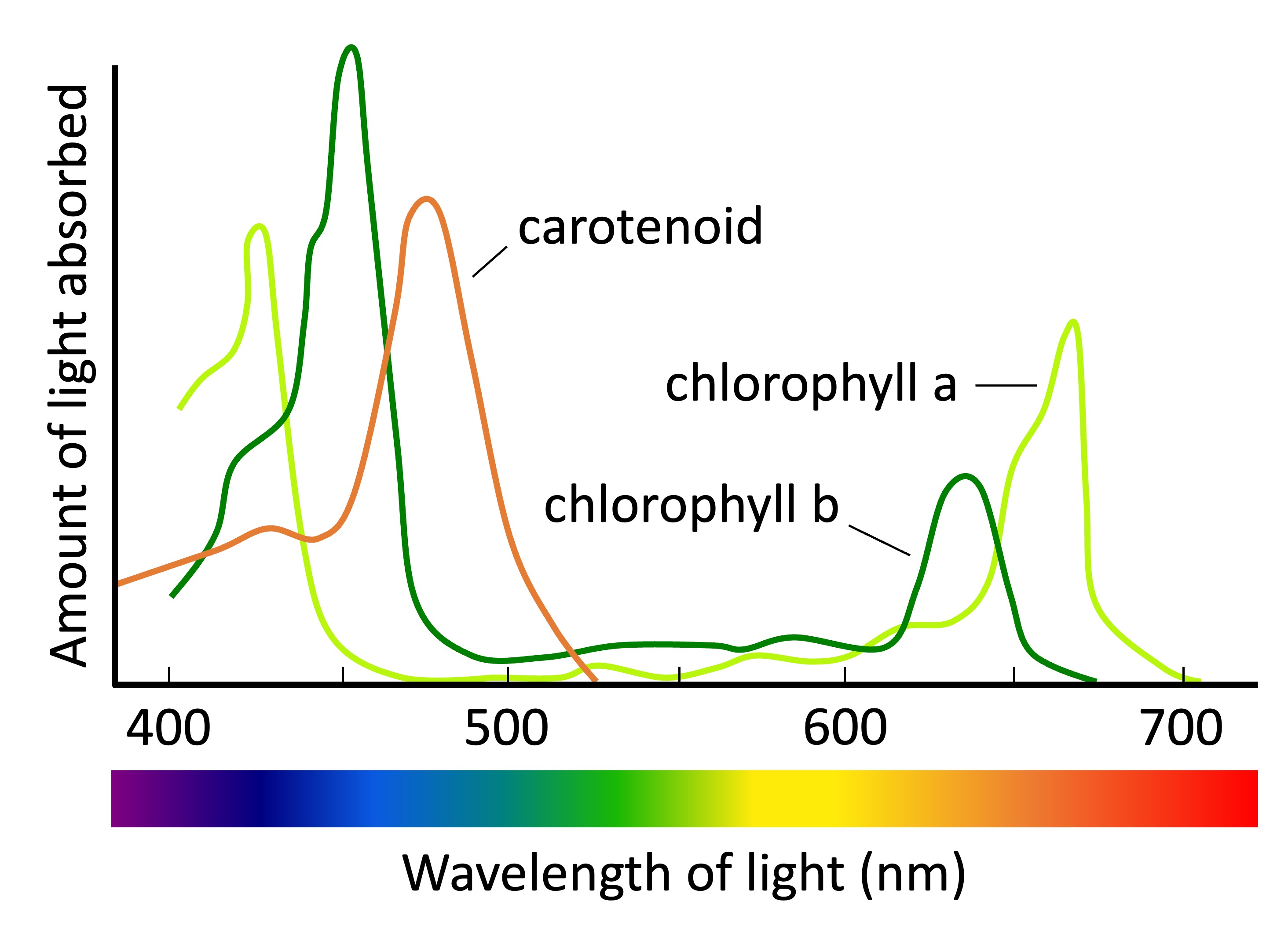
Absorption spectra (left, solid) and emission spectra (right, dotted)

Emission and Absorption Spectra: Explanation and Sample Questions

Absorption and Emission Lines
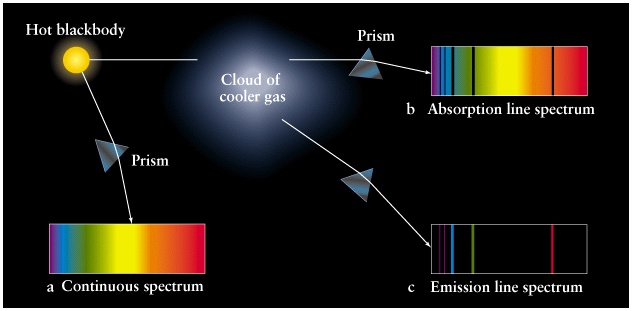
Lecture 6: Discrete Spectra of Atoms
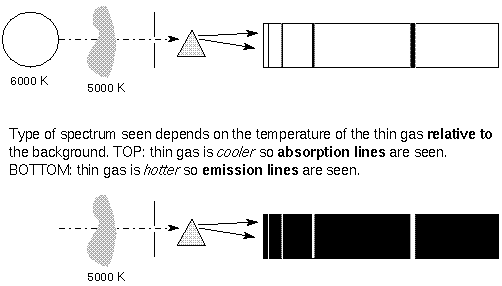
Astronomy Lecture Number 3
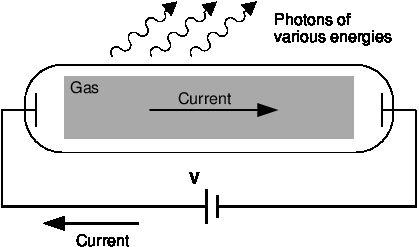
12.3 Emission and absorption spectra, Optical phenomena and properties of matter
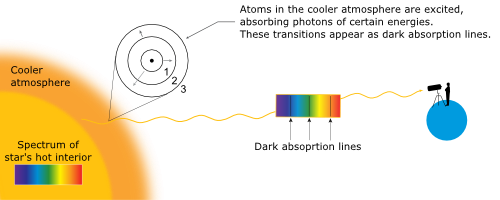
Spectral Classification - Hertzsprung-Russell Diagram - NAAP

Hydrogen emission spectrum spectroscopy successive ionisation energy patterns related to sub-shells and group of periodic table GCE A Level revision notes

Types of Spectra: Continuous, Emission, and Absorption
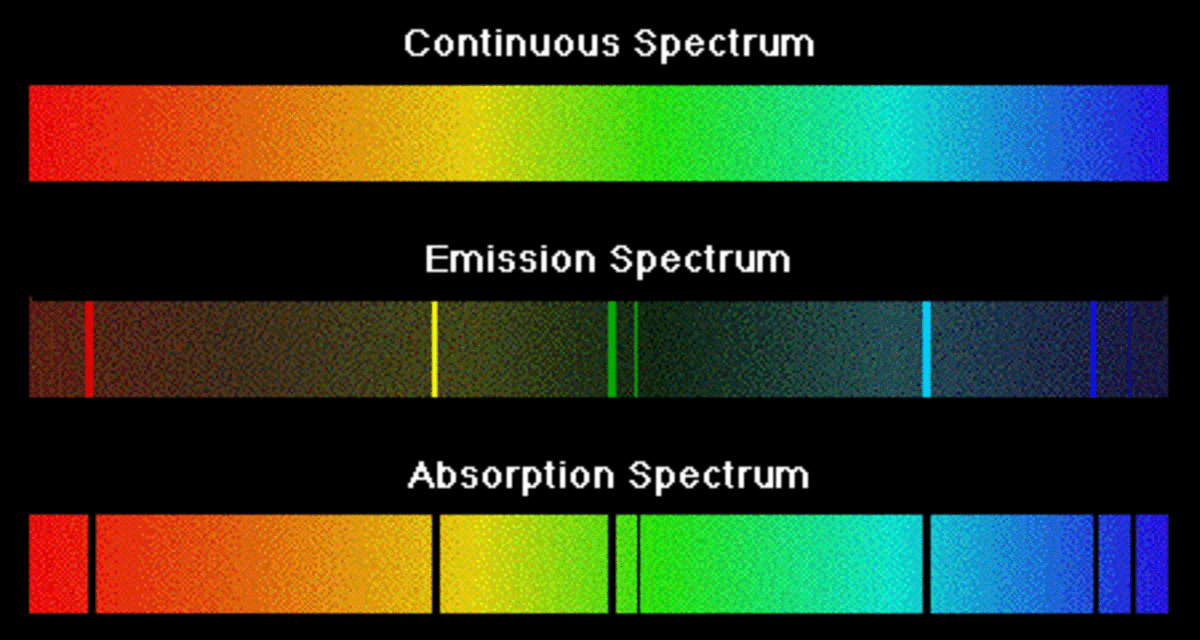
What Is The Difference Between Emission Spectra and Absorption Spectra? - HubPages